Mechanism of Enhanced Broadband Emission
Nov. 7, 2018
The lattice distortion promotes a self-trapped exciton that emits broadband white light with a very high efficiency.
Scientific Achievement
We revealed that the origin of the broadband emission in Na-doped Cs2AgInCl6 double perovskite is due to the formation of a special self-trapped exciton (STE). Pure Cs2AgInCl6 exhibits poor broadband emission due to inversion symmetry-induced parity-forbidden transition. Na-incorporation breaks the parity-forbidden transition and turns on the emission. As a result, the broadband emission photoluminescence quantum yield (PLQY) increases dramatically as the Na concentration increases. We further found that there is an optimal Na concentration to maximize the PLQY, in agreement with experimental results.
Significance and Impact
Systems with efficient and stable white emission are ideal for lighting but fundamentally difficult to achieve. Our results provide insights and design criteria for new materials that can further improve broad emission efficiency.
Research Details
- Cs2AgInCl6 exhibits poor broadband emission PLQY due to inversion symmetry-induced parity-forbidden transition.
- The PLQY can be dramatically improved via selected impurity-incorporation to break the parity-forbidden transition.
Related People
Yanfa Yan
University of Toledo
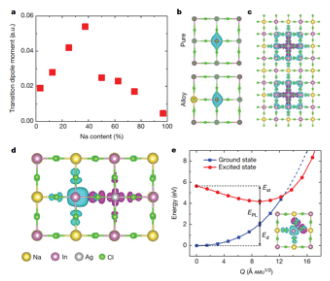
Mechanistic investigations of PLQY in Cs2Ag1−xNaxInCl6. a. Transition dipole moment as a function of Na content. b. Parity change of the electron wavefunction of the STE before and after Na incorporation. c. Configuration showing the strengthened STE confinement by the surrounding NaCl6 octahedra. d. STE in Na¬ rich. The STE is located in two neighbouring octahedra (AgCl6 and InCl6). e. Configuration coordinate diagram of the STE formation in Cs2NaInCl6 (inset). The STE is located within a single distorted InCl6 octahedron. The separation of the electron and hole makes the optical transition weak.
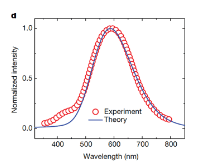
Calculated versus observed broadband photoluminescence.









